|
Molecular geometry is associated with the specific orientation of bonding atoms. It is an alkyl alcohol, a primary alcohol, a pentanol and a short-chain primary fatty alcohol. Functional Groups and Molecular Geometry. Is carboxylic acid a hydroxyl group Carboxylic acid is a class of organic compounds which contains a carbon atom which is doubly bonded with an oxygen atom and singly bonded with a hydroxyl group. It has been isolated from Melicope ptelefolia. The hydroxyl group is a functional group, alcohol is the organic compound which contains one or more hydroxyl groups attached to the alkyl group. Pentan-1-ol is an alkyl alcohol that is pentane in which a hydrogen of one of the methyl groups is substituted by a hydroxy group. 
Propanoic acid, CH3CH2COOH is a weak acid. Which functional group is present in ch3 CO ch3 c2 h4?įunctional group: Ketone name of the compound Propanone. Functional group: Ketone, name of the compound Propanone. Which functional group is present in ch3 CO ch3 c2h4?ġ Answer. Ethanol is present in the drugs and is the main component of alcoholic drinks. The alcohol that is generally used to denote the primary alcohol (i.e. The hydroxyl group (-OH) is found in the alcohol and phenol functional groups. Alcohol is an organic compound that possesses at least one hydroxyl group that is attached to the saturated carbon atom present in the alcohol molecule. What is the functional group in alcohols and phenols? They are a homologous series and have the general formula C nH 2n+ 1OH. 
What is the formula of the functional group for alcohols?Īlcohol molecules all contain the hydroxyl (-OH) functional group. It is also known as 1-propanol, 1-propyl alcohol, n-propyl alcohol, n-propanol, or simply propanol. Propan-1-ol is a primary alcohol with the molecular formula of CH3CH2CH2OH. 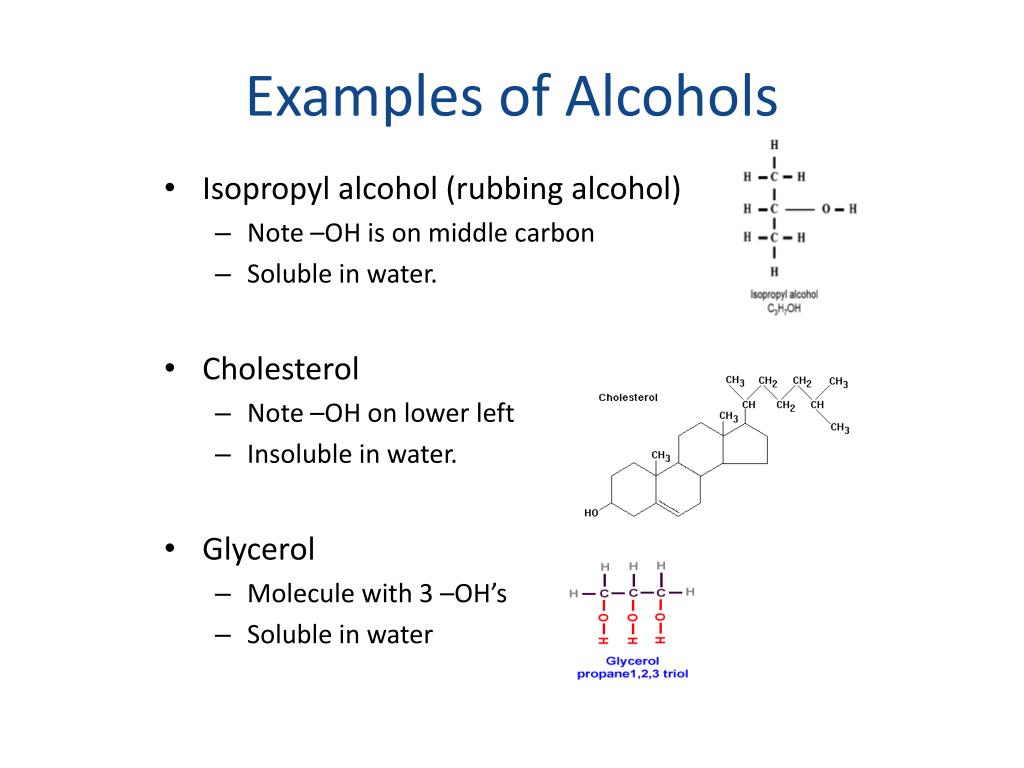
The most reactive site in an alcohol molecule is the hydroxyl group, despite the fact that the O–H bond strength is significantly greater than that of the C–C, C–H and C–O bonds, demonstrating again the difference between thermodynamic and chemical stability.What type of organic compound is CH3CH2COOH? Indeed, the dipolar nature of the O–H bond is such that alcohols are much stronger acids than alkanes (by roughly 10 30 times), and nearly that much stronger than ethers. Though they have a high pKa, alcohols are able to donate their protons in basic solutions. Alcohols can serve as weak Bronsted acids, with a pKa between 15 and 20. In organic chemistry, the term alcohol refers to a compound containing the OH (hydroxy) group. One of the most interesting properties is their acidity. Alcohol is a functional group that you are probably familiar with. Consequently, the covalent bonds of this functional group are polarized so that oxygen is electron rich and both carbon and hydrogen are electrophilic, as shown in the figure below. There are quite a few important properties that alcoholic functional groups have. In organic chemistry, the term alcohol refers to a compound containing the OH (hydroxy). The electronegativity of oxygen is substantially greater than that of carbon and hydrogen. Alcohol is a functional group that you are probably familiar with. Unlike the alkyl halides, this group has two reactive covalent bonds, the C–O bond and the O–H bond. The functional group of an alcohol is the hydroxyl group, –OH. Alcohols contain the hydroxy functional group (-OH), bonded to a carbon atom of an alkyl or substituted alkyl group. They can be prepared from and converted into many different types of compounds. 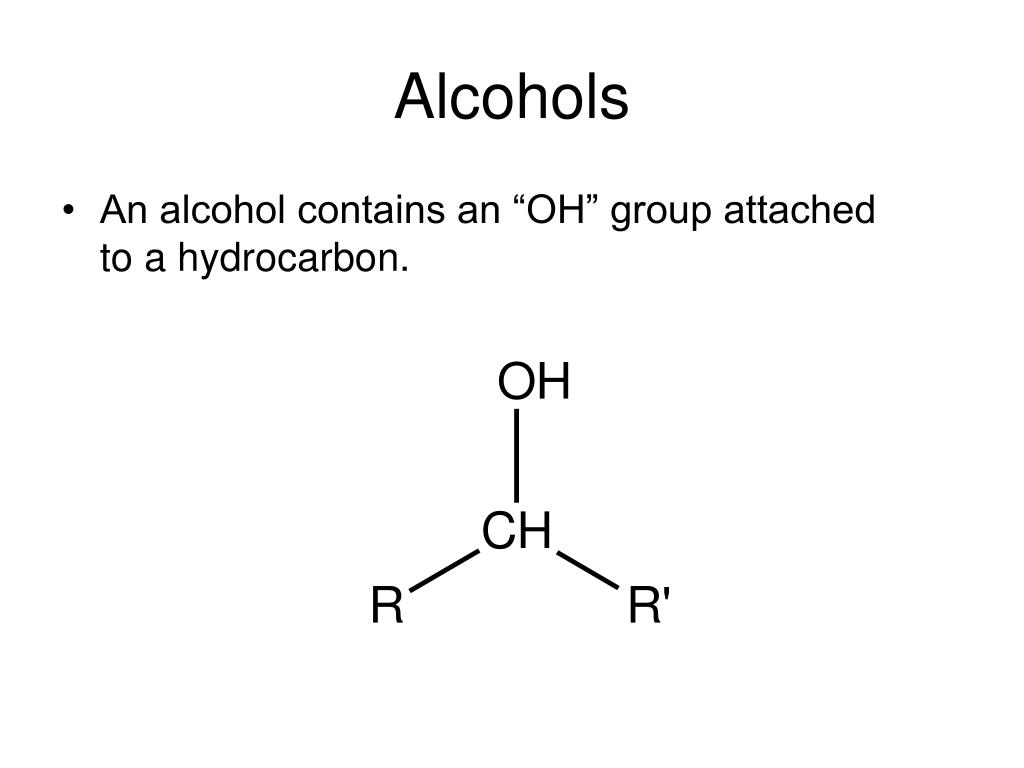
\( \newcommand\)Īlcohols are some of the most important molecules in organic chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
